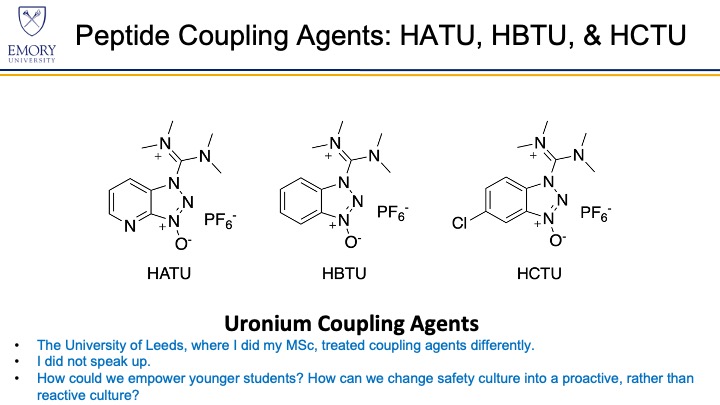
On WEDNESDAY, March 24 the CHAS Art and State of Safety Journal Club discussed the paper “Anaphylaxis induced by peptide coupling agents: Lessons learned from repeated exposure to HATU, HBTU, and HCTU.” 1st author Kate McKnelly led this discussion on this paper.The full paper can be found at this link: https://pubs.acs.org/doi/10.1021/acs.joc.9b03280. Comments on the table read are found below.

INTRODUCTION After working for years with peptide coupling agents HATU, HBTU, and HCTU[a][b],[c][d] a twenty-seven-year old female researcher (K.J.M.) developed life-threatening anaphylaxis. She began working with the aforementioned peptide coupling agents in May 2015. During the next few years, she worked heavily with these uronium peptide coupling agents. In March 2016, she began developing allergy symptoms of sneezing, coughing, and a runny nose. During the next couple of years, her symptoms progressed[e] to the point of anaphylaxis. These coupling agents are especially insidious because a severe allergy developed slowly over the course of three and a half years of exposure to the point of a life-threatening incident.
About one and a half years after beginning to work with these coupling agents, she noticed she had allergy symptoms when she weighed out coupling agents and Fmoc-protected amino acids for use in solid-phase peptide synthesis. In July[f][g] 2018, she began suspecting she was becoming allergic to coupling agents because she experienced sneezing and a runny nose immediately after spilling HCTU onto her glove. It was not until September 2018 that she experienced her first brush with allergy-induced anaphylaxis. She was at the weekly research group meeting in a seminar room down the corridor from the laboratory, and she began wheezing slightly. The wheezing was fleeting and went away after the group meeting when she left the building. A couple of weeks later, she started wheezing as she drove two labmates home. This time, the wheezing was louder—her labmates could also hear it—so she took the antihistamine diphenhydramine (generic Benadryl) to stop the reaction. Within 20 min, she could no longer hear wheezing.
Finally, in late October 2018, the researcher sat down at her desk in the lab and almost immediately began coughing, sneezing, feeling tightness in her throat, and subsequently wheezing. She attempted to remove herself from whatever she was exposed to in the lab and moved down the hallway to an office outside the lab. Once there, she continued reacting, and the wheezing progressed until she could hear a rattling wheezing sound when breathing through her nose. She immediately left the lab to obtain diphenhydramine. As[h] she exited the building, her symptoms stopped progressing. An hour after taking diphenhydramine, the wheezing subsided completely. In hindsight,[i][j][k] she should have called 911 for emergency medical help, because a throat-closing anaphylactic reaction can occur quickly, sometimes so quickly that there is barely enough time to avoid fatality.

How did this happen? How could this have been prevented?[l][m][n][o][p][q][r] We have been tackling these questions since the incident occurred. We provide this case study as a cautionary note about the potential hazards from chemical exposure that can develop over time and sneak up on a researcher. We first sought to determine what caused this anaphylactic reaction to occur. We then adjusted how peptide coupling agents were handled in the lab to minimize exposure and attempt to prevent other researchers from becoming sensitized as well. In sharing our experience here, we hope to contribute to the widespread implementation of standard operating procedures for peptide coupling agents and protect others who work with them.
LITERATURE SEARCH
We first scoured the literature for information on sensitization by peptide coupling agents HATU, HBTU, and HCTU and Fmoc-protected amino acids. Information regarding sensitization varied among chemical supplier material safety data sheets (MSDSs). HATU is reported to cause skin, eye, and respiratory irritation and is denoted by an exclamation mark hazard symbol. HBTU is reported to cause respiratory sensitization. HCTU is not reported to have known toxic effects. [s][t][u][v][w][x][y]We found only nine published cases of sensitization by the uronium coupling agents HATU and HBTU and none by HCTU or by Fmoc-protected amino acids. The first reported case implicating uronium coupling agents as chemical sensitizers came in 2003. Yung et al. described a researcher at a university that first developed eye irritation, a runny nose, and coughing (rhinitis) after weighing HBTU. Her symptoms progressed over the course of 2 weeks, developing into chest tightness, a cough, and skin rashes (urticaria) and culminating in sore, red itchy eyes, coughing, sneezing, and urticaria within 1 h of being in the laboratory. The researcher was tested with skin prick tests for allergies to HATU, HBTU, and HCTU because all chemicals were present in the lab. She tested positive for sensitivity to HATU and HBTU but negative for HCTU and various Fmoc-protected amino acids[z][aa]. Because the researcher did not exhibit sensitivity to HCTU, the authors suggested that this uronium coupling agent may be a safer alternative for widespread use. Other publications report that HCTU is nontoxic and nonirritating.

The other published instances of chemical sensitization to uronium coupling agents have involved HBTU exclusively. In 2003, another researcher, this time in a pharmaceutical plant, developed occupational rhinitis and bronchial asthma from HBTU and TBTU, which is identical to HBTU except for the counterion. The allergies were confirmed by positive skin prick and nasal challenge tests. In 2005, Bousquet et al. reported a chemistry researcher who developed allergic rhinitis and dermatitis on the hands and fingers which then progressed over the course of a year to include his face, upper back, neck, elbows, and ankles. The authors confirmed the researchers’ sensitivity to HBTU through patch testing and found he was not allergic to dimethylformamide, dichloromethane, acetonitrile, triisopropylsilane, HATU, or BOP. From 2006 to 2010, six more instances of chemical sensitization from HBTU were reported with similar respiratory and skin reactions. One example, in 2006, involved a university researcher developing an anaphylactic response to HBTU over the course of three years, similar to the case reported in this paper. All of these examples were published in allergy and other medical journals, which are not generally read by researchers who use peptide coupling agents.[ab][ac][ad][ae][af][ag]
EXPERIMENTAL CONFIRMATION
We suspected that peptide coupling agents caused K.J.M.’s allergic reactions. An allergist and clinical immunologist (W.S.) tested the researcher for allergies to a panel of over 60 allergens by skin prick tests to determine if common environmental allergens accounted for her anaphylaxis. She was only slightly allergic to two environmental allergens, but not so allergic that they would cause anaphylaxis. Skin prick tests were then performed to determine if she was allergic to HATU, HBTU, HCTU, DCC, Fmoc-leucine–OH, Fmoc-phenylalanine–OH, and Fmoc-asparagine(Trt)–OH. The researcher worked with most of the canonical amino acids in their Fmoc-protected forms, so three were chosen as representative amino acids. DCC was included as a control because it is a notorious sensitizer that the researcher had never previously worked with.
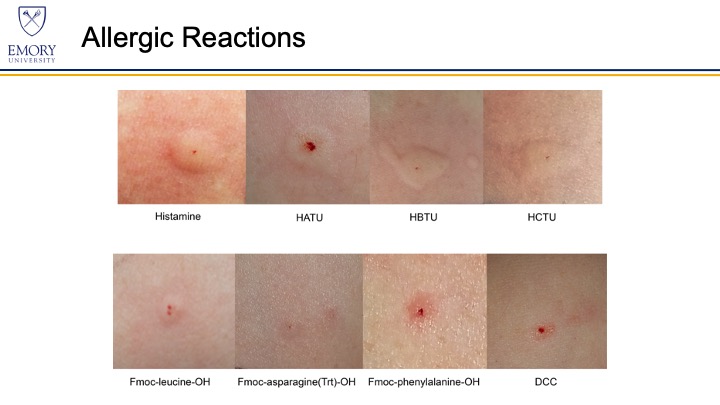
As hypothesized, the researcher had severe positive allergic reactions to uronium peptide coupling agents but only mild responses to Fmoc-protected amino acids. The coupling agents HATU, HBTU, and HCTU all caused the formation of large hives, comparable in size to those formed by the histamine positive control. DCC did not cause any reaction, which is not surprising as the researcher was never previously exposed to DCC. Fmoc-leucine–OH, Fmoc-phenylalanine–OH, and Fmoc-asparagine(Trt)–OH all elicited minor reactions and produced hives much smaller in size than the histamine positive control. The lack of a strong reaction to the Fmoc-protected amino acids is not surprising, as they are not known chemical sensitizers.
ANALYSIS
This paper serves as the first reported case of chemical sensitization resulting in anaphylaxis from three common uronium coupling agents: HATU, HBTU, and HCTU. The sensitized researcher (K.J.M.) can no longer work in her research lab. She cannot go into the building where the lab exists; the hallways, rooms, and common spaces all cause her to react, first with a runny nose and throat tightness and then with wheezing.[ah][ai][aj][ak][al][am] Her allergic response is so severe that she risks anaphylaxis whenever exposed to these coupling agents, and she now must carry an epinephrine autoinjector (generic EpiPen) as a safety precaution whenever she is near researchers actively working with peptide coupling agents. She has become sensitive to colleagues who have been in her research laboratory and must be careful to ask them to change their clothes and in some cases wash or cover their hair to prevent her exposure to the pervasive coupling agents. These events prompted the research group as a whole to re-evaluate how the group handles peptide coupling agents and to change their standard operating procedures to prevent group members from becoming sensitized to coupling agents.
Chemical sensitization causes an immune response in the form of reactions as mild as seasonal allergy symptoms, like rhinitis, and as severe as dermatitis and anaphylaxis. Many[an][ao][ap][aq][ar] chemical sensitizers are chemicals that can modify human proteins. All reactive compounds that can modify proteins should be treated as potential sensitizers unless they are known with certainty to be safe. In spite of this hazard, most researchers do not treat compounds that can react with proteins with proper precautions. Peptide coupling agents are prime examples.
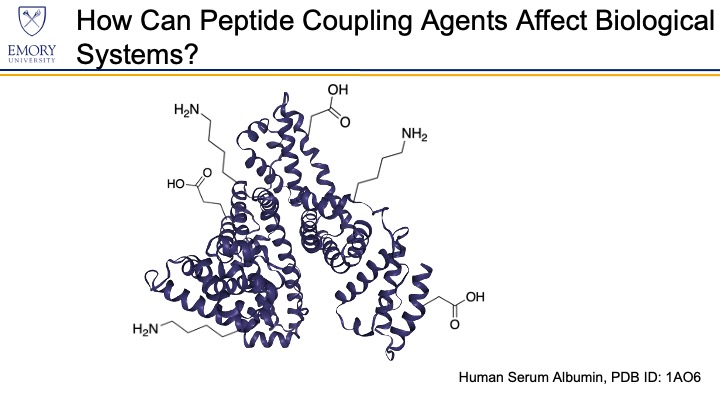
Peptide coupling agents induce the formation of an amide bond from the reaction of a carboxylic acid group with an amine group. The coupling agents react with the carboxylic acid and activate it for subsequent attack by a nucleophilic amine. After the amine reacts with the activated carboxylic acid, an amide bond forms. Human proteins display multiple carboxylic acid groups (e.g., glutamic acid and aspartic acid) and amine-containing groups (e.g., lysine) in the form of amino acid residues at protein surfaces. The reactivity of coupling agents toward amino acid residues primes them to cause sensitization by modifying proteins in the human body.
The carbodiimide coupling agent DCC (dicyclohexylcarbodiimide) is a notorious chemical sensitizer with a long history of causing sensitization. DCC was first reported as a peptide coupling agent by Sheehan and Hess in 1955. It quickly grew in popularity due to the ease with which it induced the formation of peptide bonds. Soon after its introduction, a publication reported that DCC caused three cases of allergy-induced skin rashes (contact dermatitis) in 1959. Zschunke and Folesky subsequently reported seven cases of DCC-induced contact dermatitis in a pharmaceutical plant in 1975. In 1979, two independent cases of DCC sensitivities were published in the journal Contact Dermatitis. In one case, a lab worker developed a blistering eruption rash on his hands and forearms, and in the second case, a research chemist developed a rash over nearly his entire body that persisted for five days before he was hospitalized. Since 1979, 11 more cases were reported of DCC causing similar skin contact allergic reactions. In one of these cases, the researcher also developed sensitivity to diisopropylcarbodiimide (DIC) and suffered a vesiculopapular rash on his cheeks and the backs of his hands from both DCC and DIC. The authors of each of these reported cases confirmed sensitization with skin patch tests.
The many reports of DCC sensitization lead to toxicology testing to confirm the hazard it poses to human health. DCC and DIC were nominated for testing by the National Toxicology Program in 1993. Hayes et al. then tested DCC and DIC on the skin of mice for their potential as sensitizers and in 1998 reported sensitization at concentrations as low as 0.006% (w/v) for DCC and 0.3% (w/v) for DIC. Another report in 2002 confirmed DCC and DIC as sensitizers to mice when examining the mechanism of DCC- and DIC-induced chemical sensitization. In 2011,[as][at][au] Surh et al. further characterized DCC and DIC for toxicity and carcinogenicity and determined that both DCC and DIC caused skin sensitivity in rats and mice, but only DCC exhibited carcinogenicity. The detrimental health effects of the peptide coupling agents DCC and DIC are worrisome for anyone who handles them.
HATU, HBTU, and HCTU were developed between the late 1970s and the early 2000s and are now widely used as coupling agents in peptide synthesis. Despite being implicated as sensitizers in at least ten reported cases, including the current one, they have not been rigorously tested for their immunogenic and toxicological properties.
LABORATORY ACTION PLAN
In response to the sensitization of K.J.M., we developed standard operating procedures to handle HATU, HBTU, and HCTU more safely. We found guidelines for handling sensitizers, which recommended never opening sensitizers outside of a fume hood and minimizing exposure if handling them outside of a fume hood. Our lab dedicated a portion of a fume hood to weighing out coupling agents and amino acids and placed a balance in the hood[av].[aw][ax][ay] A waste container was placed in this fume hood as a receptacle for weighing paper and other materials contaminated by coupling agents or Fmoc-protected amino acids. Coupling agents and amino acids are transferred into sealable containers before removal to individual researchers’ fume hoods. As with other standard operating procedures for handling hazardous chemicals, personal protective equipment (PPE) in the form of a lab coat, eye protection, and disposable gloves [az][ba][bb][bc][bd]should be worn at all times when handling coupling agents. We anticipate that these procedures will reduce the risk of other researchers becoming sensitized in the future.[be][bf][bg][bh][bi][bj][bk][bl][bm][bn][bo]
Any research lab that performs peptide synthesis should take extra precautions to avoid exposing researchers to coupling agents. The Supporting Information provides a standard operating procedure to handle peptide coupling agents more safely in the research laboratory by minimizing exposure[bp].
CONCLUSION
Peptide coupling agents, regardless of whether they are carbodiimide reagents, uronium reagents, phosphonium reagents, etc., all perform the same chemical function of facilitating amide bond formation and therefore can all covalently modify human proteins. If a chemical can modify human proteins, it is a prime candidate as an immune sensitizer, even if it is not a known sensitizer. We hope that our laboratory’s experience of the hazards of HATU, HBTU, and HCTU will serve as a cautionary note to those working with any peptide coupling agents.

[a]I see that PF6- is frequently the counter ion. Was this tested as an allergen?
[b]In second paragraph of the literature search part they mention a researcher who became sensitized to both HBRU and TBTU, which has a different counter ions, so while it sounds like the counter ion wasn’t tested for specifically, it doesn’t seem to be the culprit here. This makes sense since the counter ions do not partake in the coupling reaction and only has a slight influence on coupling efficiency
[c]What is the best practices to handle these coupling agents? Tilak
[d]This is discussed towards the end – also if you are interested in the protocol they shared, that is in the SI if you follow the link to the paper.
[e]Why it is important to pay special attention to unusual symptoms.
[f]and report symptoms
[g]Reporting symptoms early is also important for legal (i.e. Workers Comp) reasons
[h]Another scenario I have seen a lab worker suffer was a techinician in a electron microscopy lab. She accidently brushed her hand against a container of an epoxy they used to set up samples for the microscope and didn’t think anything of it. The next day when she can to work, her fingers started itching and keep getting worse for a week. She eventually had to leave that job.
The difference from this report is that it was a single exposure that led to the sensitization rather than repeated exposure over time.
[i]Why it is important to keep an eye on our colleagues as well and ask questions. As wild as this sounds, it is so easy for us to dismiss our own symptoms as minor even if we would be incredibly concerned about those same symptoms if we observed them in another person!
[j]A lab tech reported to me a situation in which she and a colleague were transferring insect samples between killing jars which contained 70% ethanol in the open lab. After about half an hour, she noticed that her partner was getting goofy. She then realized that they were both getting drunk from breathing the ethanol that was evaporating as they did the transfers. It’s not likely that she would have noticed this without seeing that her partner was being affected.
[k]Really good point. Also, would she have noticed her own symptoms if she had been working alone? Could’ve just interpreted this as tiredness.
[l]Would a system where researchers can report any symptoms as soon as they occur would have prevented it from getting worse?
[m]I suppose it depends on whether or not people use the system, how easy it is to use, who they are reporting to – as well as how seriously the person themselves takes their own symptoms.
[n]There are many places the someone can be exposed to allergens and the pattern they describe in the paper is more evident in retrospect than as it occurs. Animal care workers have prospective monitoring for allergies to the mice, etc. they work with, but that doesn’t prevent many from having to retire from this profession due to allergies acquired over time
[o]When I was working at the USDA, I learned of multiple people who developed allergies to moth scales over time due to a protocol in regular use that essentially required them to gently suck moths into the tip of a tube in order to move them. Gross to think about now (I never did this), but it was standard practice for a long time and many still do it this way.
[p]I have seen similar techniques outside of the chemistry lab setting. I haven’t had any lab person defend mouth-pipetting of chemicals to me since about 2005; perhaps it is a past practice, at least in academia? I’d like to think so.
[q]I knew people doing this when I was working there up through 2016!
[r]I’ve never actually seen anyone mouth pipette chemicals, so I believe the campaign against that has been a bit more effective.
[s]So “looking up the SDS” provided no information in this case.
[t]GHS SDSs should include information about sensitization, but I suspect that a chemical supplier wouldn’t add that content to a SDS based on “anecdotal evidence”. I suspect that there would need to be a published peer review study before the information was added to a SDS.
[u]Well – that is my point. We are here working on the cutting edge, but official documentation like SDSs will be necessarily behind. I cringe every time a grad student tells me “well I just looked up the SDSs and carried on” w/o having talked to ANYONE ELSE about their projects.
[v]Toxicology studies will always lag behind the introduction of new reagents. Maybe it would help to have a recognition of what classes of chemicals could be potent sensitizers and apply the precautionary principle to those. Here’s an example: “First, a chemical with dermal sensitization potential has to be able to penetrate into the skin—meaning it must have a low molecular weight, usually less than one kilodalton—and induce or elicit an immune response by being chemically reactive and electrophilic with skin proteins.” (from: https://synergist.aiha.org/201911-dermal-sensitizers) I understand that the above is broad, but it’s a start. Peptide coupling agents certainly fit the bill.
[w]It is interesting that in most cases there was little allergic reaction to HCTU, but much more severe reactions to HATU and HBTU. I wonder if once one is sensitized to the latter there is an allergic reaction to HCTU? This was the case in this study.
[x]It is also so hard to know how many people experienced these symptoms and did not connect them to exposure to these agents – so they have effectively gone unreported.
[y]I think form a chemical standpoint that would make sense. HCTU is essentially HBTU with an added chlorine, so it’s not a stretch to believe that the immune system recognizes both of these reagents in the same manner
[z]Anyone doing work with coupling reactions for peptide. peptide-mimics ought to have training on sensitizers since most of these are amines which cause sensitization
[aa]Who determines this? When I sent this article to our chemical safety specialists, they were surprised to see it! As were the members of the 1 lab I know in our building that works with these.
[ab]Another frustration with “the literature.” Safety information about chemicals doesn’t seem to have a home – it is scattered throughout so many different places that it can be easily missed by the people who need to know the information. Case in point: This case study was published in the Journal of Organic Chemistry!
[ac]These should have been posted in C&EN. That was often done during that time period as a way to alert the general chemical community. Part of the other problem is that many biochemists don’t read ACS publications.
[ad]https://cen.acs.org/safety/lab-safety/Peptide-coupling-agents-cause-severe/98/web/2020/01
[ae]It took until 202 for this to come out in C&EN?
[af]2020
[ag]While it made the rounds at the time, there are plenty of undergrads and grads working in labs who aren’t reading C&EN. C&EN is a pretty specialized resource. When I was working in a molecular genetics lab, I hadn’t even heard of C&EN.
[ah]Is there that much of the sensitizer floating around the building? Why weren’t they working with this in the hood???
[ai]Hoods are not black holes. For example, when it comes to powders they can disrupt use of the material because of the air movement in the work area
[aj]We have seen similar reports in other settings. Usually anecdotal and not as clearly documented as this. Review the literature on “multiple chemical sensitivities”. I frequently have trouble with these reports as the claims seem very wild. However, we know that sub-picomol levels of agents such as we are discussing here can induce an allergic Rx in hyper-sensitive people.
[ak]Taysir shared a comment below on why these are difficult to work with in hoods.
[al]Agree with Ralph. The appropriate engineering control for working with or weighing powders are enclosures with HEPA filtration design for that purpose, not fume hoods.
[am]Like Neal, I remember the emergence of the idea of Multiple Chemical Sensitivies and how much this confused the EHS world. There was a weird mix of science and pseudo-science that we were required to react to in addressing situations both in the lab and outside it
[an]A question this paragraph raises for me as a trainer is whether I should call attention to the chemical properties of the material the way this article does or whether I should alert people to the symptoms that they should be alert to as warning signs. The OSHA lab standard suggests training peiople on “signs and symptoms” rather than focusing on chemcials
[ao]Would it be better to do both? If one knows the symptoms but not the agent, then there could a wide range of things that could lead to these symptoms, even some not in the lab. It seems like there really needs to be causality established.
[ap]I would also think that this would be considered when discussing the design of experiments and lab protocols. You don’t want to wait until someone is having symptoms to do something about it.
[aq]I feel like the safety aspects of the research carried out in the lab doesn’t get discussed enough , even in group meetings. It’s only after something terrible has happened. I’m wondering how this culture can be affected.
[ar]Monica – that is a fundamental point of the increased interest among grad students in safety. As they move on to their careers they will become the safety leaders.
[as]1955 to 2011 – it is pretty wild to see how long it can take for a regularly used chemical to be recognized for the harm it can cause. This is important to keep in mind as we work on the cutting edge of scientific experimentation!
[at]Part of the problem, again, may be in the communication forums used during that time. I wonder if some of the more common social media will make this easier now…
[au]I may not be following the correct social media, I don’t see a lot of lab procedure information there. Where would one look for these stories?
[av]The measures taken are pretty basic – and involve things that are now available in virtually ALL labs. This is another important consideration.
[aw]Is there a process for decontaminating the balance and hoods in place? Can the agents be deactivated by other chemicals?
[ax]I believe bleach will not work with these chemicals, may be cleaning by ethanol is the best solution
[ay]We generally use methanol followed by water
[az]Since some of the agents are known to cause respiratory distress, would a face mask or any type of respiratory protection help?
[ba]We have been using N95 masks when working with these reagents. I don’t have any data to support this practice though
[bb]This will need fit tested
[bc]Yup, DEHS at my institution provides that testing
[bd]I suspect that the allergeric reactions could be triggered by skin exposure and other environmental contamination as much as respiratory exposure. The NP95 masks will help by avoiding cross contamination from your hands to your face, which may be helpful
[be]As a result of this paper, I convinced my group to buy a new balance to keep in the hood for weighing out HCTU and to follow the suggested protocol. The issue we ran into is that due to the hood flood it takes a very long time for the balance to tare. Since our peptide synthesizer is cartridge based we have to weigh the coupling reagent individually for each amino acid. As a result, it now takes days instead of hours to finish weighing all the amino acids. Many researchers in our lab have instead elected to wear N95 masks when weighing the coupling reagent instead of using the hood balance
[bf]That is interesting. We didn’t have any hoods in my lab with balances in them, but I have used them in other labs and had no issues with them taring. I’m wondering now what made the difference.
[bg]Probably due to our hoods being ancient to be honest. I am not happy about this solution, but I understand why people go for it
[bh]generally it is difficult to weigh powders inside the chemical fume hood due to air flow, however, crystalline material is ok
[bi]Housekeeping is very important to handle such chemicals in the lab
[bj]It also takes some time and practice to get used to working with powders and crystalline materials that I don’t think most students really get until they are working in a research lab. Working a hood does add to the complexity of this.
[bk]The lab I worked in in the 80’s had ventilation weighing station for working with silca and asbestos dusts. It takes a very careful ventilation design for sensitive balances to be able to operate in a wind current. We also had a special table which was very heavy to provide a steady surface for the balance.
[bl]A weighing enclosure will also work for this purpose
[bm]I agree with Jessica about the practice/ or hands-on on weighing
[bn]We just use a three side piece of acrylic around the scale and have no problems. As an alternative, you could teach people in the lab to weigh out chemicals using analytical subtractive techniques. This is the fastest method by far. (weigh out your vial, add some chemical to the vial in the hood, put the lid on and weigh again, add solvent to desired concentration)
[bo]Was working only with solutions considered? I.e., upon receipt of a new bottle of coupling agent, dissolve it in a solvent to a known concentration, then for each use, volumetrically measure what’s needed and then dilute? Wet methods are excellent for controlling exposures to dusts/particulate. Look at construction sites during large-scale demo…you usually see a big hose running to minimize dust.
[bp]We use a synthesizer often. All chemicals are weighed out and diluted in the hood. The sealed bottles are then transferred to the synthesizer. A tube is then use to carry any escaping vapors back to the fume hood.


Thank you for the great article. I have worked extensively with this uronium coupling reagents for last 8 years. After Kate’s report, I finally figured out the reason of my 8 years of itching and rashes. I finally stopped taking citrizine. Thank you Kate.